Dan Benesh - Personal Site

Contact
Humboldt University of Berlin
Molecular Parasitology
Philippstr. 13, Haus 14
10115 Berlin, Germany
Email address: dbenesh82@gmail.com
Education and Employment
Group leader - 2017-2022
Humboldt University of Berlin
Research focus: Constraints on parasite life cycles
DFG Fellowship - 2013-2017
University of California, Santa Barbara
Research focus: Parasite life cycles in the food web
Postdoctoral researcher - 2007-2013
Max Planck Institute for Evolutionary Biology, Plön, Germany
Research focus: The evolution of complex parasite life cycles
M.S. and Ph.D - 2004-2007
University of Jyväskylä, Finland
Thesis topic: Larval life history strategies of an acanthocephalan parasite
B.S. - 2000-2004
University of Nebraska, Lincoln, U.S.A.
Major: Biological Sciences, Minors: Philosophy and Entomology
My CV and my profiles on ResearchGate, Google Scholar, Publons, LinkedIn, and GitHub

Research Interests
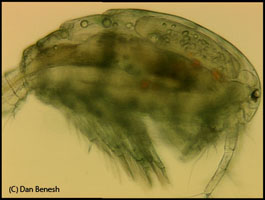
I am interested in the evolutionary ecology of parasites, mainly helminths (i.e. worms). My research has covered diverse topics, from parasitic manipulation of host behavior to parasite community ecology. But a central focus is the evolution of complex parasite life cycles, particularly two questions: 1) why have complex life cycles evolved and 2) how do parasites adapt to complex life cycles? I use both cross-species comparative analyses and experiments to explore these questions.
Why have a complex life cycle?
Many parasites, including some of medical and veterinary importance, obligatorily use several hosts in their life cycle, i.e. they have a complex life cycle. At first glance, complex life cycles are puzzling, because a simple, one-host life cycle seems intuitively easier to complete. Parasites also must adapt to multiple host physiologies and immune systems. However, there are potential advantages for parasites with a complex life cycle, such as avoiding mortality from predation, achieving higher transmission rates, reaching a larger size at maturity, and increasing the probability to find a mate.
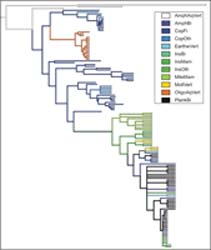
Broad, cross-species comparisons are one way to test these presumed benefits. From 2014 to 2016, I worked on a DFG-funded project to compile life cycle information for parasitic helminths. The resulting database summarizes decades of research and includes nearly 1000 species of thorny-headed worms, tapeworms, and roundworms. Using these data, we found that parasites with longer life cycles infected conspicuously smaller first hosts (which probably leads to higher propagule transmission) and slightly larger final hosts (which enables in larger, more fecund reproductive sizes). Another analysis showed that complex life cycle helminths are generalists (i.e. infect more host species) when they have the opportunity, but this generalism comes at the cost of reduced growth. More simply, the database documents the incredible diversity in parasite strategies – the gif below shows how parasite size changes over the life cycles of different species. Some of the factors underlying this diversity can be explored in an interactive chart I built.

How to adapt to a complex life cycle?
How should complex life cycle parasites divide their growth and development among their multiple hosts? Life history models suggest that the size and age at which parasites are transmitted between hosts should maximize lifetime growth while minimizing mortality. Consistent with this, independent helminth taxa that are transmitted along similar trophic links (e.g. copepod – fish or insect – terrestrial vertebrate) have frequently converged on similar life history strategies, presumably because they experience similar growth and mortality rates.
But how might adaptations in one life stage (like larvae in the intermediate host) affect other life stages (like adults in the definitive host)? Do parasites use the same mechanisms in multiple hosts or can each stage evolve independently in response to different selection pressures? Since 2017, I have been at the Humboldt University in Berlin, where we have a grant to study the microevolutionary constraints on parasite life cycles. We are comparing transcriptomes from parasite stages with similar functions (e.g. growth) but in different hosts (intermediate vs definitive host). Less overlap in gene expression between hosts would imply decoupling – parasites use different genes in different hosts to do similar things. This transcriptomics project is being led by PhD student Laura Gramolini.
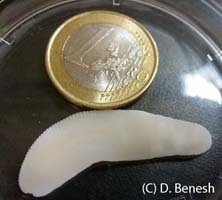
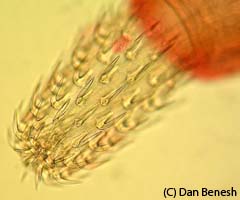
One of the focal species of our transcriptomics project is the tapeworm Schistocephalus solidus. It has three hosts in its life cycle (a copepod, a stickleback, and a piscivorous bird). The entire life cycle of S. solidus can be conveniently run in the laboratory, because the bird final host can be replaced with an in vitro system. The ability to breed worms allows us to perform studies that are impossible for most parasites, such as parentage and quantitative genetic studies. Some findings from my postdoc at the MPI in Plön include: high variation in paternity after indirect competition for fertilizations, significant non-genetic parental effects on larval performance, and strong lifetime inbreeding depression and its potential to be purged through continued inbreeding. Through experimental S. solidus infections, we discovered that big worms in copepods are better at infecting sticklebacks. In many parasites, growth in intermediate hosts may increase the likelihood of establishing infection in the next host, as we observed this trend in a larger comparative analysis. The comparative approach has also yielded insights into Schistocephalus’s unique life history strategy, like why it refrains from growth in birds (because they undergo massive growth in fish).

Publications
Check out my citation statistics here. Do not hesitate to contact me for pdfs.
Benesh, D.P. 2023. Selection on an extreme-yet-conserved larval life-history strategy in a tapeworm. Evolution. doi: 10.1093/evolut/qpad034.
Benesh, D.P., Chubb, J.C., and Parker, G.A. 2022. Adaptive division of growth and development between hosts in helminths with two-host life cycles. Evolution. 76 (9): 1971-1985. doi: 10.1111/evo.14574.
Benesh, D.P., Chubb, J.C., Lafferty, K.D., and Parker, G.A. 2022. Complex life-cycles in trophically transmitted helminths: Do the benefits of increased growth and transmission outweigh generalism and complexity costs? Current Research in Parasitology & Vector-Borne Diseases: 100085. doi: 10.1016/j.crpvbd.2022.100085.
Phillips, J.A., Vargas-Soto, J.S., Pawar, S., Koprivnikar, J., Benesh, D.P., and Molnár, P.K. 2022. The effects of phylogeny, habitat and host characteristics on the thermal sensitivity of helminth development. Proceedings Royal Society B: 20211878. doi: 10.1098/rspb.2021.1878.
Benesh, D.P., Parker, G.A., and Chubb, J.C. 2021. Life-cycle complexity in helminths: what are the benefits? Evolution 75 (8): 1936-1952. doi: 10.1111/evo.14299.
Froelick, S., Gramolini, L., and Benesh, D.P. 2021. Comparative analysis of helminth infectivity: growth in intermediate hosts increases establishment rates in the next host. Proceedings Royal Society B 288: 20210142. doi: 10.1098/rspb.2021.0142.
Benesh, D.P., Parker, G.A., Chubb, J.C., and Lafferty, K.D. 2021. Tradeoffs with growth limit host range in complex life cycle helminths. The American Naturalist 197 (2): E40-E54. doi: 10.1086/712249.
Chubb, J.C., Benesh, D.P., Parker, G.A. 2020. Ungulate helminth transmission and two evolutionary puzzles. Trends in Parasitology 36: 64-79. doi: 10.1016/j.pt.2019.10.010
Benesh, D.P. 2019. Tapeworm manipulation of copepod behaviour: parasite genotype has a larger effect than host genotype. Biology Letters 15: 20190495. doi: 10.1098/rsbl.2019.0495
Erin, N., Benesh, D.P., Henrich, T., Samonte, I.E., Jakobsen, P.J., Kalbe, M. 2019. Examining the role of parasites in limiting unidirectional gene flow between lake and river sticklebacks. Journal of Animal Ecology 88: 1986-1997. doi: 10.1111/1365-2656.13080
Benesh, D.P. 2019. Crowding in the first intermediate host does not affect infection probability in the second host in two helminths. Journal of Helminthology 93 (2): 172-176. doi: 10.1017/S0022149X1800007X
Benesh, D.P., Lafferty, K., and Kuris, A. 2017. A life cycle database for parasitic acanthocephalans, cestodes, and nematodes. Ecology 98 (3): 882. doi: 10.1002/ecy.1680
Benesh, D.P. 2016. Autonomy and integration in complex parasite life cycles. Parasitology 143: 1824-1846. https://doi.org/10.1017/S0031182016001311 (invited review)
Benesh, D.P. and Kalbe, M. 2016. Experimental parasite community ecology: intraspecific variation in a large tapeworm affects community assembly. Journal of Animal Ecology 85 (4): 1004–1013. doi: 10.1111/1365-2656.12527
Hafer, N. and Benesh, D.P. 2015. Does resource availability affect host manipulation? – an experimental test with Schistocephalus solidus. Parasitology Open 1: e3. doi: http://dx.doi.org/10.1017/pao.2015.3
Tuomainen, A., Valtonen, E.T., and Benesh, D.P. 2015. Sexual segregation of Echinorhynchus borealis (Acanthocephala) in the gut of burbot (Lota lota). Folia Parasitologica 62: 061. doi: 10.14411/fp.2015.061
Benesh, D.P., Aura, R.-L., Andersin, A.-B., and Valtonen, E.T. 2015. The occurrence of Echinorhynchus salmonis Müller, 1784 in benthic amphipods in the Baltic Sea. Folia Parasitologica 62: 052. doi: 10.14411/fp.2015.052
Aura, R.-L., Benesh, D.P., Palomäki, R., and Valtonen, E.T. 2015. The natural history of Echinorhynchus bothniensis Zdzitowiecki and Valtonen, 1987 (Acanthocephala) in a high Arctic lake. Folia Parasitologica 62: 051. doi: 10.14411/fp.2015.051
Benesh, D.P., Chubb, J.C., and Parker, G.A. 2014. The trophic vacuum and the evolution of complex life cycles in trophically-transmitted helminths. Proceedings of the Royal Society B. 281: 20141462. doi: 10.1098/rspb.2014.1462
Benesh, D.P., Weinreich, F., Kalbe, M., and Milinski, M. 2014. Lifetime inbreeding depression, purging, and mating system evolution in a simultaneous hermaphrodite tapeworm. Evolution 68 (6): 1762-1774. doi: 10.1111/evo.12388
Weinreich, F., Kalbe, M., and Benesh, D.P. 2014. Making the in vitro breeding of Schistocephalus solidus more flexible. Experimental Parasitology 139: 1-5. doi: 10.1016/j.exppara.2014.02.002
Andreou, D. and Benesh, D.P. 2014. Copulation order, density cues, and variance in fertilization success in a cestode. Parasitology 141 (7): 934-939. doi:10.1017/S0031182014000043
Benesh, D.P. 2013. Parental effects on the larval performance of a tapeworm in its copepod first host. Journal of Evolutionary Biology 26 (8): 1625-1633. doi: 10.1111/jeb.12165
Henrich, T., Benesh, D.P., and Kalbe, M. 2013. Hybridization between two cestode species and its consequences for intermediate host range. Parasites & Vectors. 6: 33. doi: 10.1186/1756-3305-6-33
Benesh, D.P., Chubb, J.C., and Parker, G.A. 2013. Complex life cycles: why refrain from growth before reproduction in the adult niche? The American Naturalist 181: 39-51. doi: 10.1086/668592
Weinreich, F., Benesh, D.P., and Milinski, M. 2013. Suppression of predation on the intermediate host by two trophically-transmitted parasites when uninfective. Parasitology 140: 129-135. doi: 10.1017/S0031182012001266
Benesh, D.P. and Hafer, N. 2012. Growth and ontogeny of the tapeworm Schistocephalus solidus in its copepod first host affects performance in its stickleback second intermediate host. Parasites & Vectors 5: 90. doi: 10.1186/1756-3305-5-90
Benesh, D.P., Weinreich, F., and Kalbe, M. 2012. The relationship between larval size and fitness in the tapeworm Schistocephalus solidus: bigger is better? Oikos 121: 1391-1399. doi: 10.1111/j.1600-0706.2011.19925.x
Benesh, D.P., Chubb, J.C., and Parker, G.A. 2011. Exploitation of the same trophic link favors convergence of larval life history strategies in complex life cycle helminths. Evolution 65: 2286-2299. doi: 10.1111/j.1558-5646.2011.01301.x
Benesh, D.P. 2011. Intensity-dependent host mortality: what can it tell us about larval growth strategies in complex life cycle helminths? Parasitology 138: 913-925. doi: 10.1017/S0031182011000370
Benesh, D.P. 2010. What are the evolutionary constraints on larval growth in a trophically-transmitted parasite? Oecologia 162: 599-608. doi: 10.1007/s00442-009-1507-6.
Benesh, D.P. 2010. Developmental inflexibility of larval tapeworms in response to resource variation. International Journal for Parasitology 40 (4): 487-497. doi: 10.1016/j.ijpara.2009.10.001
Benesh, D.P., Seppälä, O., and Valtonen, E.T. 2009. Acanthocephalan size and sex affect the modification of intermediate host coloration. Parasitology 136 (8): 847-854. doi: 10.1017/S0031182009006180
Hasu, T., Benesh, D.P., and Valtonen, E.T. 2009. Differences in parasite susceptibility and cost of resistance in naturally exposed and unexposed host populations. Journal of Evolutionary Biology 22 (4): 699-707. doi: TT10.1111/j.1420-9101.2009.01704.x
Benesh, D.P., Hasu, T., Seppälä, O., and Valtonen, E.T. 2009. Seasonal changes in host phenotype manipulation by an acanthocephalan: time to be transmitted? Parasitology 136 (2): 219-230. doi: 10.1017/S0031182008005271
Seppälä, O., Valtonen, E.T., and Benesh, D.P. 2008. Host manipulation by parasites in the world of dead-end predators: adaptation to enhance transmission? Proceedings of the Royal Society B 275: 1611-1615. doi: 10.1098/rspb.2008.0152
Benesh, D.P., Valtonen, E.T., and Seppälä, O. 2008. Multidimentionality and intra-individual variation in host manipulation by an acanthocephalan. Parasitology 135 (5): 617-626. doi: 10.1017/S0031182008004216
Benesh, D.P., Kitchen, J., Pulkkinen, K., Hakala, I., and Valtonen, E.T. 2008. The effect of Echinorhynchus borealis (Acanthocephala) infection on the anti-predator behavior of a benthic amphipod. Journal of Parasitology 94 (2): 542-545. doi: 10.1645/GE-1380.1
Benesh, D.P., Valtonen, E.T., and Jormalainen, V. 2007. Reduced survival associated with precopulatory mate guarding in male Asellus aquaticus (Isopoda). Annales Zoologici Fennici 44 (6): 425-434.
Benesh, D.P. and Valtonen, E.T. 2007. Effects of Acanthocephalus lucii (Acanthocephala) on intermediate host survival and growth: implications for exploitation strategies. Journal of Parasitology 93 (4). 735-741. doi: 10.1645/GE-1093R.1
Benesh, D.P. and Valtonen, E.T. 2007. Proximate factors affecting the larval life history of Acanthocephalus lucii (Acanthocephala). Journal of Parasitology 93 (4). 742-749. doi: 10.1645/GE-1094R.1
Benesh, D.P. and Valtonen, E.T. 2007. Sexual differences in larval life history traits of acanthocephalan cystacanths. International Journal for Parasitology 37 (2): 191-198. doi: 10.1016/j.ijpara.2006.10.002
Benesh, D.P., Hasu, T., Suomalainen, L.R., Valtonen, E.T., and Tiirola, M. 2006. Reliability of mtDNA in an acanthocephalan: the problem of pseudogenes. International Journal for Parasitology 36 (2): 247-254. doi: 10.1016/j.ijpara.2005.09.008
Benesh, D.P., Duclos, L.M., and Nickol, B.B. 2005. Behavioral response of amphipods harboring Corynosoma constrictum (Acanthocephala) to various components of light. Journal of Parasitology 91 (4): 731-736. doi: 10.1645/GE-440R.1